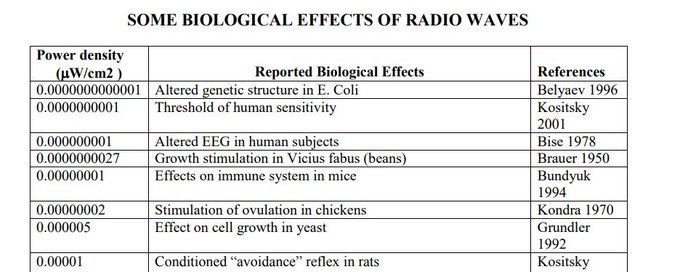
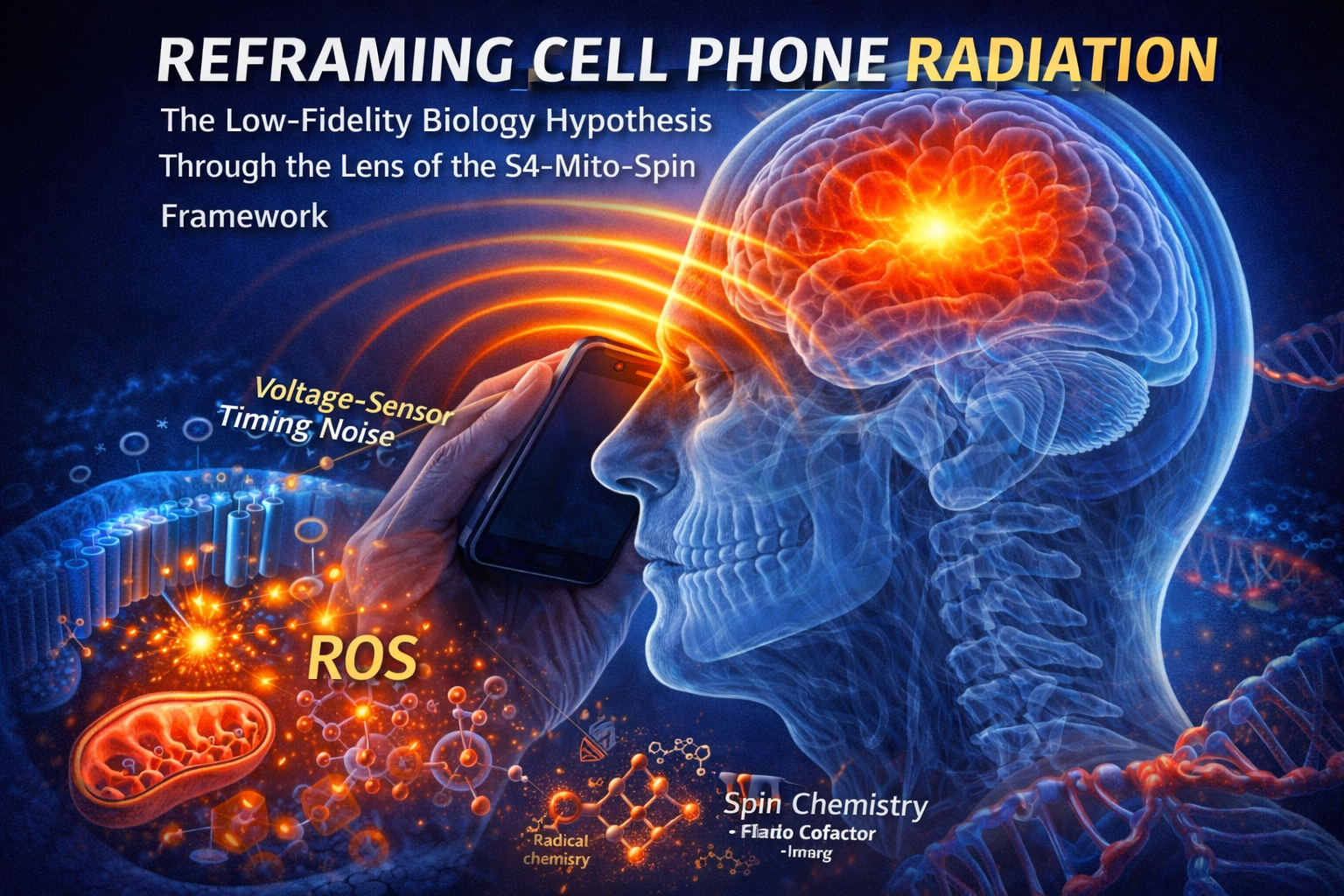
Why the April 14 Cell paper materially strengthens RF Safes S4MitoSpin framework
The strongest mechanistic objection to non-thermal EMF biology has always been the same: where is the transducer? The April 14, 2026 Cell paper, Electromagnetic field-inducible in vivo gene switch for remote spatiotemporal control of gene expression, just put a real candidate on the table. The paper identifies Cyb5b as an essential mediator of an EMF-inducible gene switch and shows that externally applied EMF input can be converted into rhythmic oscillatory calcium dynamics that control gene expression in vivo.
That is not a minor detail. It is a major shift in the argument.
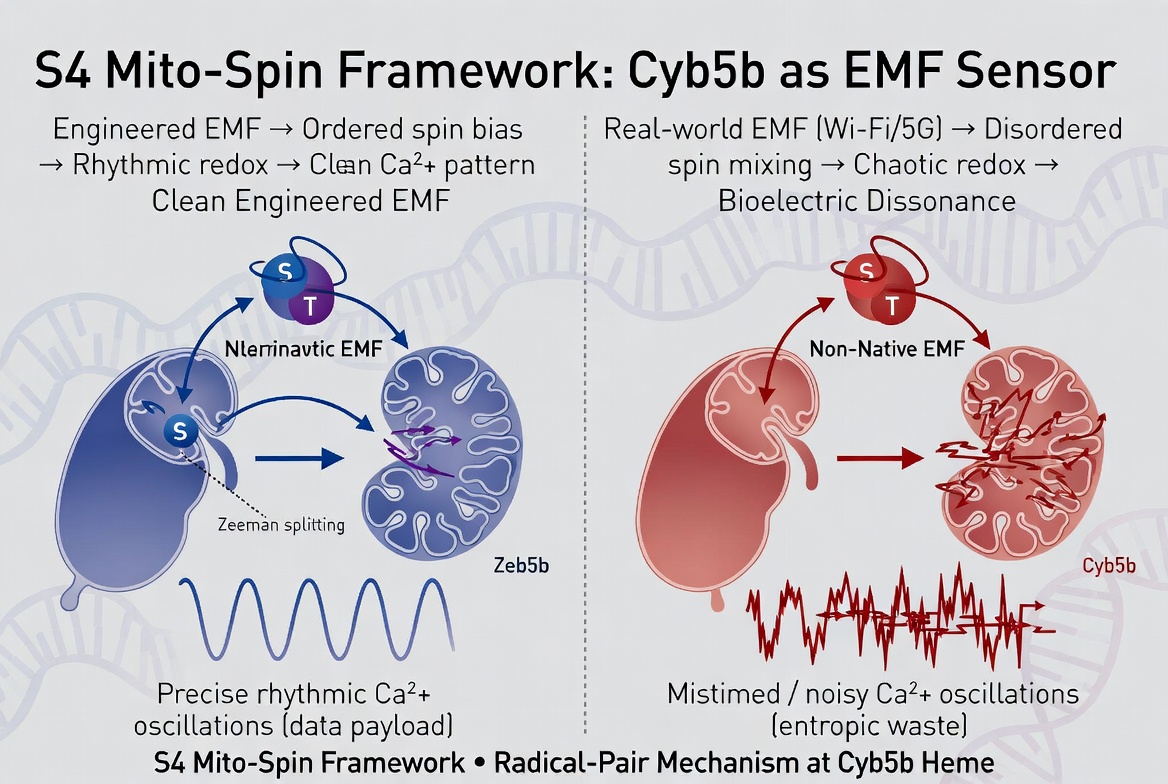
For years, the default dismissal of weak-field biology has been that there is no established mechanism below heating thresholds. RF Safes S4MitoSpin framework has argued the opposite: that non-thermal EMFs couple into biology through three vulnerable layers at once S4 voltage-sensor timing, mitochondrial and NOX redox amplification, and spin-sensitive chemistry in heme and flavin systems. The new Cyb5b paper does not merely fit that framework. It strengthens its most contested pillar: Spin.
The paper that changed the map
What did the Cell team actually show?
They built an EMF-inducible in vivo gene switch with precise spatiotemporal control. A CRISPR-Cas9 screen identified cytochrome b5 type B (Cyb5b) as an essential mediator, likely acting as the EMF sensor. The switch was not turned on by a blunt calcium flood. It was turned on by rhythmic oscillatory calcium dynamics rather than generic calcium influx, which is exactly why the result matters so much. The output was not a trivial reporter either: EMF activation drove OSK-based partial reprogramming in aged mice, conditional expression of mutant human APP with Alzheimers-like pathological features, and Tph2 expression that restored serotonergic activity and improved depressive-like behaviors in a mouse depression model.
That is a complete signal chain:
external EMF Cyb5b-dependent intracellular transduction patterned calcium code controlled gene expression organism-level phenotype
Once that chain exists in the literature, the old weak EMFs cannot possibly talk to living systems in a specific way line is finished.
Why Cyb5b matters so much
Cyb5b is not some vague quantum placeholder. It is a real, named, biologically important protein: CYB5B / cytochrome b5 type B, also known as OMB5 or CYB5-M. It is an outer mitochondrial membrane hemoprotein and an electron carrier. In other words, it sits exactly where an RF Safe-style mechanism would want a sensor to sit: at a membrane interface where redox chemistry, mitochondrial signaling, and calcium handling can all talk to each other.
That location matters more than almost any abstract argument ever has. The outer mitochondrial membrane is not just packaging. It is a live signaling surface. It sits at the front line of contact-site biology, calcium crosstalk, and redox state control. If you were going to look for a biologically plausible EMF transducer that could convert field exposure into a meaningful intracellular update, an outer-membrane hemoprotein is a far more serious candidate than the cartoon version of mystery energy.
Cyb5b also comes with real biochemical credentials. A 2024 Cell Reports paper on CYB5A and CYB5B showed that mitochondrial CYB5B, not CYB5A, is required for the sterol-C4 oxidation step in cholesterol biosynthesis in HeLa cells. That study also described CYB5B as anchored to the outer membrane of mitochondri, with most of the protein exposed to the cytosol. So the new Cell paper is not elevating an obscure protein with no prior biological footprint. It is repurposing a real mitochondrial redox component as a field-facing control node.
The hidden headline is calcium pattern, not calcium quantity
The single most important sentence in the new paper may be the one most people will skate past: the EMF-inducible switch was activated by rhythmic oscillatory calcium dynamics rather than generic calcium influx.
That one phrase should reframe the entire discussion.
Cells do not treat calcium as a mere more or less variable. Calcium is a code. Frequency matters. Timing matters. Phase matters. Duty cycle matters. Oscillation structure matters. In excitable tissues, endocrine tissues, immune cells, and differentiation pathways, cells read calcium as structured information. A clean oscillatory program does one thing; a mistimed or distorted calcium pattern does something else. RF Safes own framework centers this exact point: timing errors at the membrane become distorted Ca waveforms, and those distorted waveforms become downstream biological error.
So the deepest significance of the Cyb5b paper is not simply that EMFs can affect cells. It is that EMFs can write intracellular timing structure. They can push biology at the level where cells decide what a signal means.
That is the bridge RF Safe has been arguing for all along. Not heat. Not brute-force damage. Control-layer interference.
Why this is a major win for the Spin pillar
Now bring in the second line of evidence: Andrew Yorks protein magnetofluorescence work.
York and colleagues showed that modest laboratory magnetic fields can modulate the brightness of GFP-like fluorescent proteins in the presence of appropriate cofactors. They demonstrated this at room temperature and body temperature, in vitro, in bacteria, and even in cultured mammalian cells with the construct targeted to mitochondria. The early GFP-family response was small, but it was real. They also showed that purified mScarlet plus an FMN photoproduct could produce a larger response, and their later MagLOV engineering update pushed the effect dramatically higher.
That matters because it demolishes the old caricature that room-temperature protein spin sensitivity is too fragile, too exotic, or too speculative to matter in biology. It is already happening in protein systems under biologically relevant conditions.
Just as important, Yorks team explicitly points to the radical-pair mechanism as the standard explanatory framework for magnetoresponsive proteins and fluorophores. They were appropriately careful not to overclaim that their own observations alone prove the full model, but the direction of travel is unmistakable: protein-scale magnetoresponse is real, engineerable, and consistent with spin-dependent chemistry.
And the case only became stronger after Yorks initial work. A 2025 JACS paper on mScarlet3 developed a quantitative mechanism for a giant magnetic-field effect in a red fluorescent protein system and tied the effect to a triplet spin-correlated radical pair involving the protein and flavin chemistry. Another 2025 report, Magnetic resonance control of reaction yields through genetically-encoded protein:flavin spin-correlated radicals in a live animal, showed that RF magnetic fields can influence radical-pair reaction yields in biologically relevant conditions and even inside a living animal.
That is the context in which the Cyb5b result lands. The Spin pillar is no longer carrying the burden of mere plausibility. It now stands on a growing experimental base showing that protein-associated radical chemistry can be biased by externally applied fields and turned into measurable biological output.
RF Safes mechanistic extension: from heme spin bias to calcium code
This is where RF Safes S4MitoSpin framework becomes especially powerful.
Cyb5b is a heme protein on the outer mitochondrial membrane. That makes it an ideal candidate for a spin-selective redox gate. RF Safes mechanistic extensionis direct: EMF exposure perturbs electron-transfer timing and spin-state branching at or near the Cyb5b heme center; that altered redox state is then converted into a structured calcium signal at the mitochondrial surface; the calcium pattern is then decoded by the cell into transcriptional state. Put more simply: spin bias redox bias calcium pattern gene expression. That is exactly the kind of transduction chain RF Safe has been pointing to.
The S4 pillar explains how weak polarized fields can inject timing noise into voltage-gated channels. The Mito pillar explains how distorted calcium and voltage patterns get amplified into oxidative stress and state change in mitochondria-rich tissues. The Spin pillar explains how heme- and flavin-rich systems can be hit even when classical S4 gating is not the main entry point. The Cyb5b paper does not replace that architecture. It gives it a newly credible mitochondrial front-end sensor.
From clean oscillations to dirty noise
What the Cell group built was a clean signal path. Their system used a tuned EMF input to generate a bio-orthogonal calcium pattern and then harnessed that pattern for specific payloads.
That is precisely why the safety implications are so serious.
A clean, engineered field can produce a clean intracellular code. But biology in the real world is not surrounded by clean, single-purpose signals. It is surrounded by non-native, pulsed, polarized, multiplexed, duty-cycled, amplitude-modulated field environments. In everyday technological settings, the cell is not receiving one elegant instruction. It is receiving a cluttered stream of timing-rich perturbations. Within the S4MitoSpin framework, the logical expectation is clear: if clean EMFs can write clean calcium codes through real cellular hardware, then dirty EMFs can write dirty codes. Not purposeful remote control, but corrupted control. Not a therapeutic command, but a mistimed one. Not a meaningful biological update, but an entropic one.
That is what RF Safe means by low-fidelity biology.
Low-fidelity biology is not a movie-scene catastrophe. It is an increased biological error rate. It is the wrong calcium burst at the wrong time. It is the wrong redox flare during a repair decision. It is the wrong transcriptional state during a developmental or immune checkpoint. It is the wrong pulse timing in a tissue that lives and dies by timing. Over days, months, and years, that becomes what RF Safe has called Bioelectric Dissonance: a widening gap between the code a cell should be reading and the code it actually receives.
And once biology is understood at that level, the language of average SAR starts looking much less complete. The relevant variable is no longer just absorbed power. It is signal structure and timing fidelity.
Why the ceLLM / photonic-redox control plane matters here
This is also why RF Safes expanded manuscript, Toward a Topology-Gated Photonic-Redox Control Plane, suddenly feels much closer to the laboratory than to speculation.
That manuscript argues that mitochondria are not merely energy factories; they are multiscale signaling hubs where redox chemistry, calcium, membrane potential, mtDNA topology, hydration-shell physics, and organelle contact sites can convert weak perturbations into stored state updates. It explicitly frames the problem as one of cross-scale frequency conversion: fast optical or electromagnetic events are converted into lower-frequency electrochemical envelopes that the cell can amplify, integrate, and remember.
The new Cyb5b result plugs directly into that architecture. The manuscript already proposed that organelle contact sites amplify weak perturbations into canonical signaling variables like Ca2+, membrane potential, and retrograde signaling. The Cell paper now provides a plausible EMF-facing hardware element at the outer mitochondrial membrane that can sit upstream of exactly that kind of calcium control.
In other words, the ceLLM manuscript was looking for a serious transducer layer. Cyb5b now looks like one of the strongest candidates on the board.
The fit gets even tighter when you look at adjacent mitochondrial evidence. A 2026 ACS Nano paper on frequency-selective terahertz irradiation reported that 34.5 THz, but not a nearby tested frequency, enhanced mitochondrial biogenesis through calcium influx and the PGC-1NRF1/2TFAM axis. That does not prove RF Safes full model by itself. What it does show is that mitochondria are not passive bystanders in field biology. They are responsive, frequency-sensitive control hardware.
The next experiments are obvious now
Once Cyb5b is on the table, the right experiments are no longer hard to imagine.
Mutate the heme coordination environment of Cyb5b and ask whether the EMF-induced calcium program collapses.
Shift the static magnetic background and ask whether the calcium response curve moves with it.
Alter hyperfine coupling through isotope substitution and ask whether sensitivity changes.
Compare the engineered waveform from the Cell system with messy, real-world mixed waveforms and measure not just calcium amplitude, but calcium entropy, jitter, phase stability, ROS timing, transcriptional noise, and repair fidelity.
That is the crucial shift. The key metric is no longer simply did calcium go up? The key metric is how faithfully the signal was timed. RF Safes manuscript is already explicit that the framework has to live or die by falsifiable predictions. The Cyb5b result makes that test program sharper, not softer.
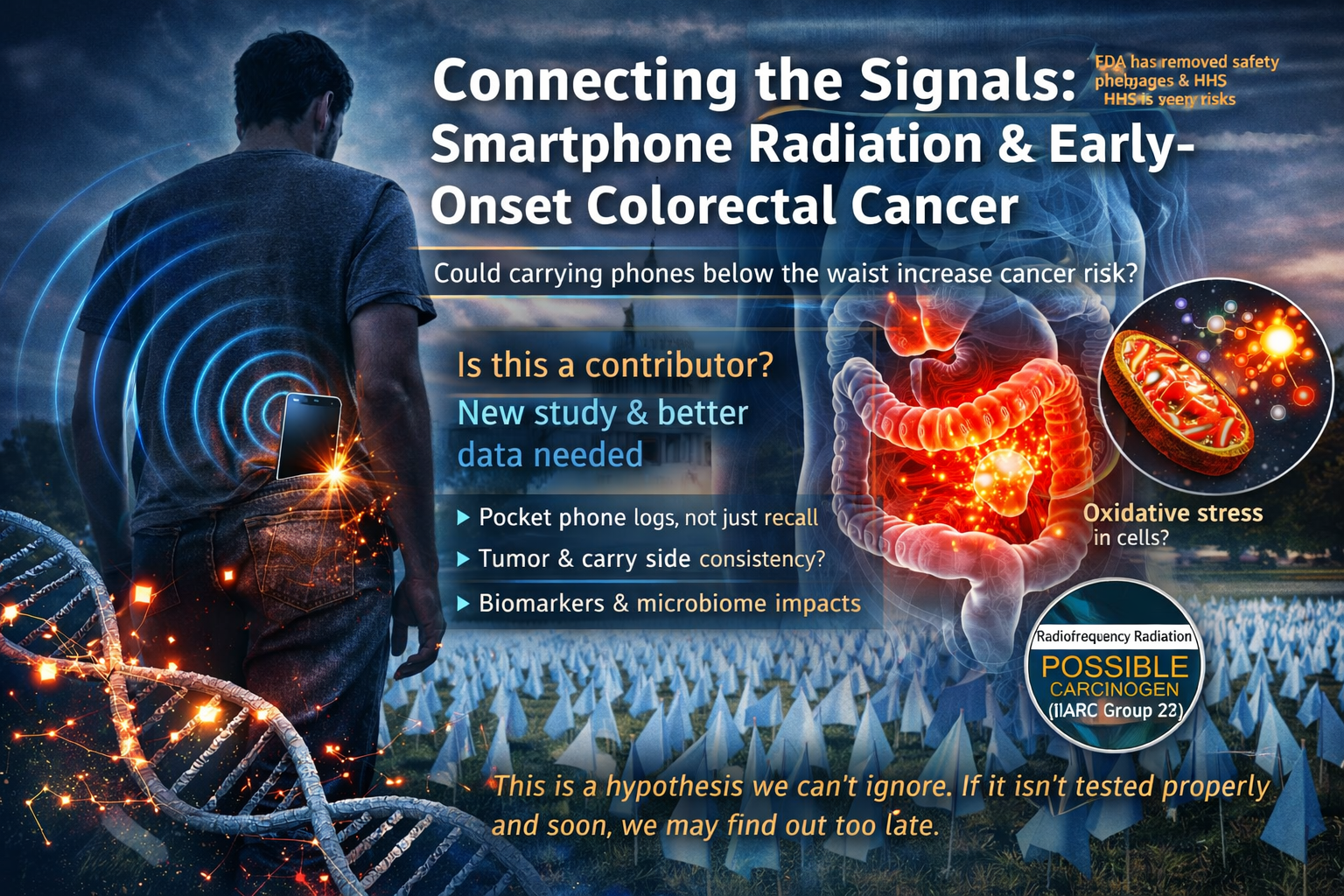
What this means for EMF risk, design, and policy
The practical meaning is not subtle.
Once biology is known to contain real EMF-facing transducers capable of converting external field structure into intracellular timing codes, the old heat-only safety story becomes dramatically less adequate. The biologically important variables now plainly include waveform, pulsing, phase, timing, tissue architecture, mitochondrial density, redox load, and spin-sensitive cofactors. That is exactly the direction RF Safe has been pushing in its S4MitoSpin framework, its Clean Ether Act blueprint, and its broader evidence hub.
It also clarifies why RF Safe keeps arguing for a transition toward light-first and wired-first indoor networking. If chronic high-duty-cycle RF exposure is capable of degrading biological timing fidelity one mistimed calcium burst at a time, then the rational engineering response is not panic. It is better infrastructure.
Higher-fidelity biology demands higher-fidelity environmental design.
The bottom line
The April 14 Cell paper did not merely add another headline to the EMF debate. It delivered three things the field badly needed:
a real mitochondrial transducer candidate in Cyb5b,
a mechanistic bridge in rhythmic calcium coding,
and whole-animal functional outputs showing that the signal chain can reach phenotype.
Yorks magnetofluorescence work, and the follow-on radical-pair literature around fluorescent proteins and RF-controlled reaction yields, already showed that spin bias in proteins under modest external fields is real. The new Cyb5b paper now anchors that general principle to a biologically important mitochondrial membrane protein with direct access to calcium signaling and gene control.
Put together, these findings materially strengthen the Spin component of RF Safes S4MitoSpin framework and make the problem of low-fidelity biology under non-native EMF exposure far more concrete.
The old question was whether weak fields can couple to biology.
The new question is how much error chronic dirty exposures inject into the codes life uses to stay coherent.